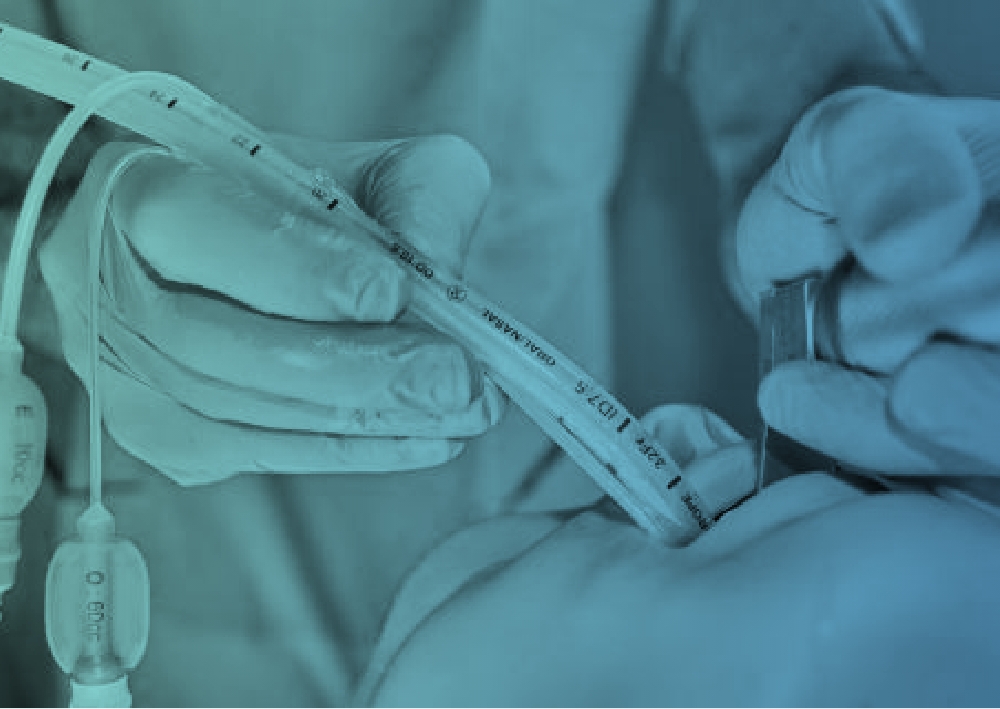
A simple solution to a complex problem
Spritztube® is a patented medical device by Med Europe s.r.l. that revolutionizes the approach to airway management. It was developed to overcome the limitations of traditional devices, which require removal of the supraglottic airway to proceed with endotracheal intubation.
Its distinctive feature is the ability to function both as a supraglottic airway for immediate ventilation, and as an endotracheal tube for definitive intubation, simply by modifying the cuff configuration.
Spritztube® is available in disposable version (medical grade PVC with embedded metal coil) and resterilizable version (medical grade silicone with embedded metal coil), representing the concrete answer to the daily challenges of anesthesiologists, intensivists, and emergency operators.
Versions
- Disposable (medical grade PVC with embedded metal coil)
- Resterilizable (medical grade silicone with embedded metal coil)
Materials
- Tube – medical grade PVC with embedded metal coil
- Balloons – distal in medical grade PVC, proximal in PUR
- Cuffs – medical grade PVC
- Stylet – PVC coated malleable steel
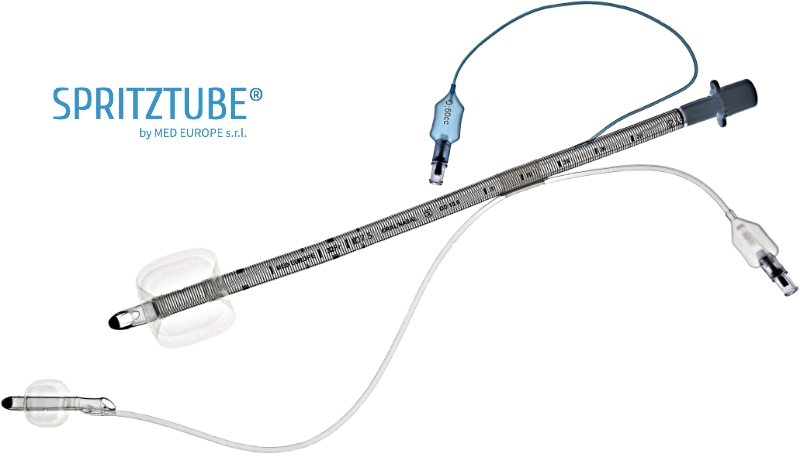
Spritztube® anatomy
Every component is designed to ensure safety, effectiveness, and ease of use.
Reinforced PVC/Silicone tube
Anti-kinking structure.
Proximal cuff
Supraglottic for ventilation, tracheal in case of intubation.
Esophageal cuff
Protects airway from gastric contents.
Malleable stylet with non-traumatic tip
Keeps cuffs in line during introduction, serves as guide during intubation.
Flexible tab
Links the two cuffs while keeping them independent.
Two dedicated inflation lines
Independent control of cuffs.

Why choose Spritztube®
Da presidio sovraglottico a tubo endotracheale in pochi, semplici passaggi.Insert
Spritztube® blindly or with laryngoscope. Inflate both cuffs. Patient is ventilated and protected.
Vale
Calmly, while patient breathes. Decide if to intubate and how. No time pressure, better decisions.
Intubate
Deflate proximal cuff. Advance into trachea with laryngoscope or fiberscope. No need to exchange airway devices.
Ventilate
Inflate tracheal cuff. Intubation completed. Same device, new function.
Spritztube® certifications
Why Spritztube® is different
A direct comparison highlighting the advantages in terms of time, safety, and simplicity.
| Traditional | Spritztube® |
|---|---|
| 6 step | 5 step |
| 2 devices | 1 device |
| Mandatory exchange | No change |
| Apnea during exchange | Continuous ventilation* |
| Extra maneuvers | Reduced maneuvers |

Learn more
Clinical benefits
Discover all the benefits for anesthesiologists, emergency, and healthcare facilities.
Read benefitsTecnichal datasheet
Materials, certifications, dimensions, and ordering information.
Download the datasheetAvailable Download
Product brochure, Technical datasheet, Instructions for use and IFU (PDF).
Learn more